0.21 Wilson Score
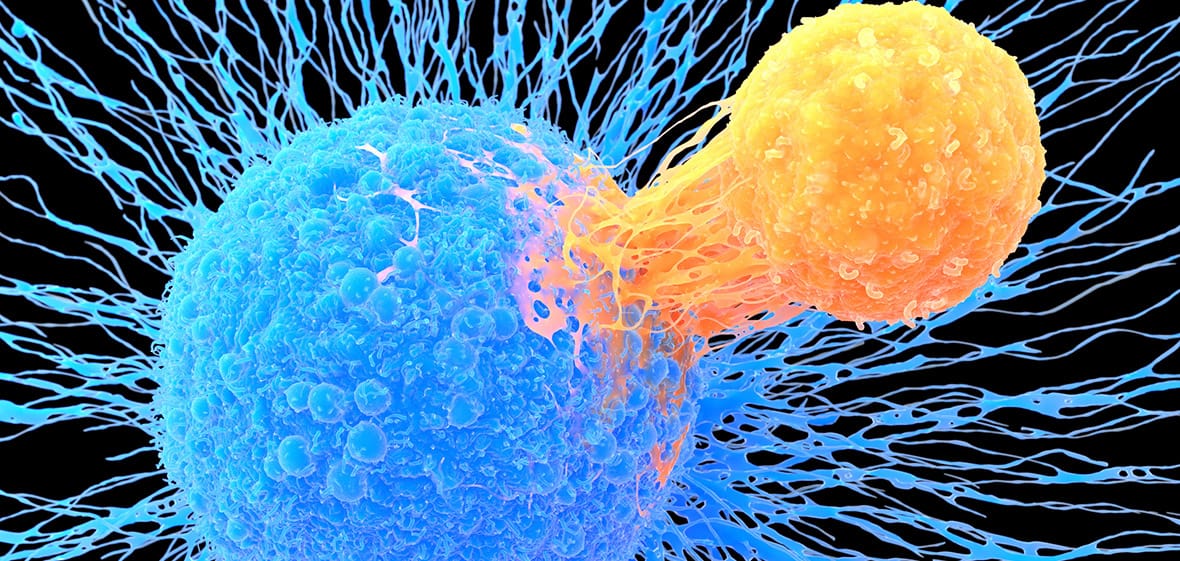
‘Jumping gene’ enzyme can make big, precise changes to human DNA
Improved editing tool inserts gene-size sequences into human genome at specific locations, broadening potential treatments
Introduction overview
Thanks to CRISPR and related gene-editing techniques, scientists are getting better and better at making small changes to our DNA to treat certain diseases. But they are still working out how to insert long stretches of DNA at precise locations in the human genome—a feat that could expand the range of conditions treatable with editing. Now, a research team has increased the efficiency of a set of CRISPR-related molecular tools borrowed from bacterial cells. They report today in Science that they’ve used those tools to install segments of DNA the size of whole genes in human cells. The result could make it easier to turn this bacterial system into practical disease treatments, scientists say, although some formidable technical challenges loom.
“There’s been huge interest in putting in large pieces of DNA,” says immunologist Dimitrios Wagner of Baylor College of Medicine, who has been developing methods to edit the genomes of immune cells and wasn’t connected to the study. The work “is an important step toward making the technology better.”
A technique for plugging in hefty chunks of DNA at specific locations could speed development of therapies for illnesses, such as cystic fibrosis and hemophilia B, that result from a variety of mutations. More than 2000 DNA defects have been implicated in cystic fibrosis, for instance. If researchers could insert a whole corrected gene, they could craft a single therapy for many mutations. They would not have to devise, test, and gain regulatory approval for a custom treatment that addressed each genome glitch, like the personalized fix reported earlier today for a baby with an enzyme disorder. Gene insertion technologies might also allow scientists to improve chimeric antigen receptor–T cells, genetically altered immune cells that fight cancer.
The challenge has been how to do it. Gene therapy often relies on tame viruses to carry full-size genes into cells. But the approach is imprecise. The added genes land at random locations in the genome, or may not enter it at all. In contrast, precision is the hallmark of gene-editing techniques such as CRISPR—which has been approved for treating sickle cell anemia and is being tested against a range of other illnesses—and base editing, a CRISPR spinoff that swaps out troublesome DNA bases and has also entered clinical trials. Such techniques work well for changes of less than 200 bases, but human genes are typically thousands of bases long. Another limitation of traditional CRISPR systems is that they leave breaks in both DNA strands at the editing site, which can lead to harmful effects such as loss or gain of genetic material.
The new work instead relies on enzymes called CRISPR-associated transposases, or CASTs, that can insert DNA without creating these double-stranded fractures. In bacterial cells, CASTs help parasitic DNA sequences known as transposons hop from place to place in the genome. A guide RNA strand in the enzymes pinpoints where to insert the nomadic sequences.
Previous studies have determined that CASTs are nearly 100% efficient at shoehorning hunks of DNA into target locations within bacterial genomes. But they’ve fallen short in human cells. Last year, for example, biochemist Samuel Sternberg of Columbia University and colleagues reported that the efficiency of the process was only about 0.1%.
To improve the approach, Sternberg’s lab combined forces with a team led by genome editing virtuoso David Liu, a chemical biologist at Harvard University and the Broad Institute. It’s hard to design improvements to a system as complex as a CAST, so the team turned to an artificial evolution system containing bacteria and their viral parasites that Liu’s lab had developed. The researchers inserted the sequences for some CAST proteins into the genomes of the viruses. For the pathogens to reproduce, their CASTs had to manipulate the DNA of the bacteria, activating a gene in the microbes that enables viral replication.
Randomly mutating the viruses provided fodder for natural selection to evolve strains whose CAST proteins were most efficient at making shifts and insertions in their hosts’ DNA. The approach accelerates the speed of natural evolution by 1000 times, Liu says. “This was what we used to crack the problem.”
After letting the CAST components evolve for hundreds of viral generations, the researchers fine-tuned them and then put the system through tests in a variety of human cell lines. The experiments showed the updated system worked about 200 times better than the original, with efficiencies that typically fell between 10% and 30%. The researchers also showed in human cells that the upgraded CASTs could make changes that might be clinically relevant, such as inserting a gene for a blood-clotting protein known as factor IX, which is faulty in hemophilia B.
The paper provides the first demonstration that CASTs can insert large pieces of DNA “at appreciable levels in human cells,” and it “plants a flag” for the efficiency of the approach, says molecular geneticist Shondra Miller of St. Jude Children’s Research Hospital. Stanley Qi, a bioengineer at Stanford University, praised the team’s “impressive work in directed evolution of key protein components.”
The study “is a tour de force in terms of engineering,” says gene therapy researcher Luigi Naldini of San Raffaele University School of Medicine, but “there’s a way to go for clinical translation.” CASTs are relatively large, which could make it hard to get them into patient cells. Delivering CASTs to the right tissues in the body could also be difficult. And Liu notes that researchers may have to contend with cells’ natural defenses against pathogens. “Many cells object to having large pieces of DNA inserted into them. They think they are under viral attack.”
Scientists are developing other methods for adding big pieces of DNA to the human genome, including a technique known as PASSIGE that Liu and colleagues unveiled in 2021, which relies on a DNA-swapping enzymes called recombinases instead of CASTs. “It’s too early to say what’s going to be the best technology,” Sternberg says. But now researchers “have the opportunity to compare them head-to-head.”
Related papers
What did you think of this post?
Like
1